Gain new perspectives for faster progress directly to your inbox.

Autoimmune diseases, those where a person’s immune system attacks its cells, include serious conditions like multiple sclerosis (MS), type 1 diabetes, and celiac disease. These diseases have significant impacts on people’s lives and currently have no cure, and incidents are rising across the globe. Studies suggest a connection between COVID-19 infection and an increased risk of developing autoimmune diseases, which makes treating them more urgent.
Scientists are pursuing a new therapy called inverse vaccines to combat these diseases and other autoimmune conditions. What is an inverse vaccine? It’s a treatment that teaches the immune system to recognize its antigens as safe and to stop attacking them.
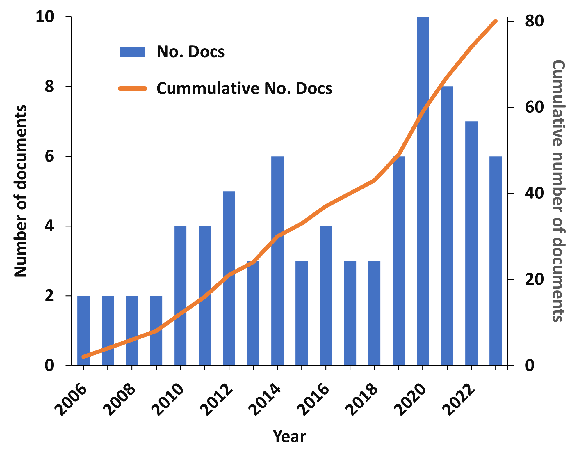
According to our recent analysis of the CAS Content CollectionTM, the largest human-curated repository of scientific information, research on inverse vaccines is in its nascent stages but growing quickly (see Figure 1). For people living with MS, type 1 diabetes, celiac disease, and other conditions like allergies, inverse vaccines could be the breakthrough they need.
How do inverse vaccines work?
Inverse vaccines, also called tolerogenic vaccines, introduce the body's proteins (autoantigens) in a specific way to induce tolerance, which means that the immune system learns to recognize the autoantigens as harmless.
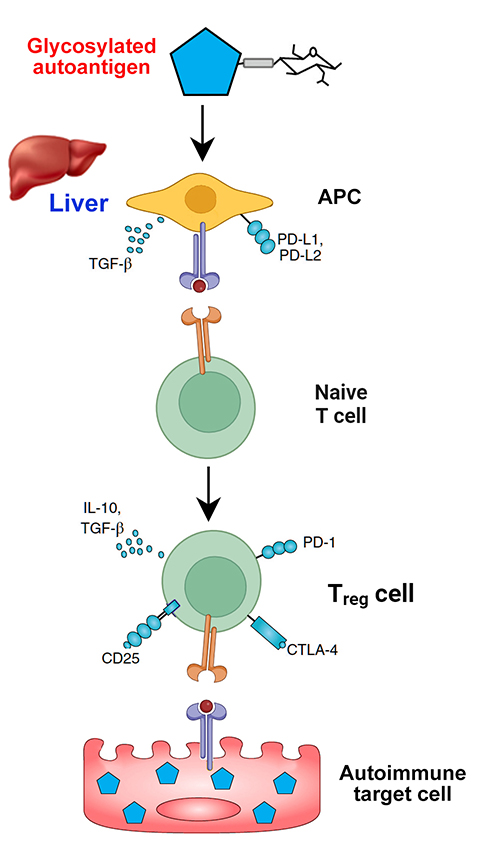
This treatment has been researched for the past couple of decades, but a recent breakthrough technique was reported last year. A phenomenon known as peripheral immune tolerance, which is carried out in the liver, ensures that immune reactions do not take place in response to every damaged cell in the body. It has been discovered that tagging molecules with a sugar known as N-acetylgalactosamine (pGal) could mimic this process, sending the molecules to the liver where tolerance has been acquired.
By attaching pGal to the target antigen, the inverse vaccine gets the liver to recognize that antigen as “safe” (Figure 2). This tricks the specialized liver cells into teaching the immune system T cells to tolerate and protect the target antigen rather than attack it. In mouse models of a multiple sclerosis-like disease, the inverse vaccine completely reversed the autoimmune attack and restored proper nerve function.
Researchers are exploring different methods to deliver these vaccines, including microparticles, nanoparticles, or engineered viruses, to ensure they reach their target effectively. Protein antigens can be encapsulated into porous PLGA (poly(lactic-co-glycolic acid)) microspheres using methods like "self-healing encapsulation" to achieve sustained antigen release. The PLGA delivery system can help protect the antigens and facilitate their targeted delivery to induce immune tolerance. Antigens and other immunomodulators can also be efficiently loaded onto the surface of PLGA microparticles by conjugating them with short peptides. This surface conjugation approach can help retain the integrity of the antigens during the encapsulation process.
Before encapsulation into PLGA microparticles, protein antigens can first be loaded into polysaccharide (dextran) glassy particles through freezing-induced phase separation. This two-step process can help improve the loading efficiency and stability of the antigens.
In addition to PLGA microparticles, some other drug delivery systems that have been explored for inverse vaccines include:
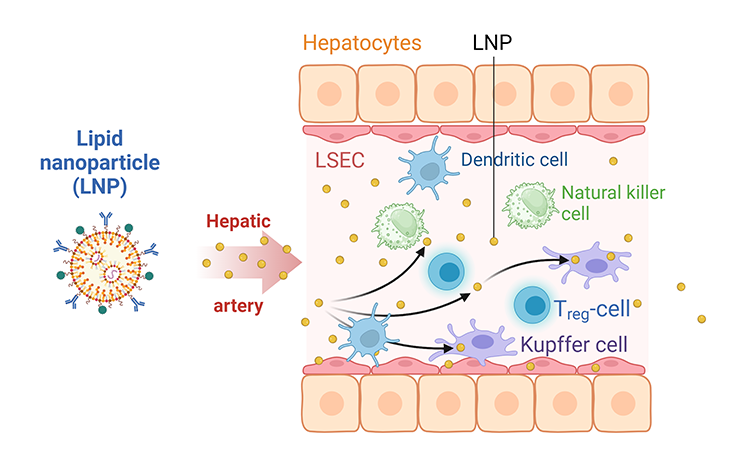
- Liposomes: Lipid-based nanoparticles that can encapsulate and deliver vaccine antigens and adjuvants (see Figure 3).
- Emulsions: Options include oil-in-water emulsions that can serve as vaccine delivery platforms and have been used to enhance the immune response.
- Virus-like particles (VLPs): These non-infectious, self-assembling nanoparticles derived from viral structural proteins can present antigens in a native-like formation.
- Polymer-based nanoparticles: Biodegradable polymers like chitosan and poly(lactic acid) have been explored for nanoparticle-based vaccine delivery. They could be engineered to target the liver and induce immune tolerance for inverse vaccines.
- Exosomes: These naturally occurring nanoparticles derived from cell membranes can be engineered to carry vaccine antigens and adjuvants, and their inherent targeting and immune-modulating properties make them a promising platform for inverse vaccine delivery.
Benefits of inverse vaccines
The key innovation behind inverse vaccines is their ability to selectively remove the memory of the immune system for specific self-antigens that it mistakenly attacks in autoimmune diseases. They do this by hijacking natural tolerance-teaching mechanisms of the liver, rather than broadly suppressing the entire immune system. Current treatments for autoimmune diseases focus on suppressing the immune system to reduce inflammation and slow disease progression. However, this can leave patients more vulnerable to infections and other complications.
Instead, inverse vaccines aim to be highly targeted. They ideally suppress only the specific immune response attacking a particular autoantigen (the body's protein mistakenly targeted for destruction). This can lead to fewer side effects like increased susceptibility to infections, more effective treatment by targeting the root cause of the autoimmune reaction, reduced treatment burden, and better quality of life.
The best-case scenario is that inverse vaccines reprogram the immune system to permanently ignore the autoantigen, leading to long-term remission or even a cure for some diseases. For patients with MS or type 1 diabetes, these are powerful breakthroughs that could potentially cure these devastating diseases. Allergies and allergic asthma are also promising targets for inverse vaccines. There is potential in using inverse vaccines for people who undergo an organ transplant, teaching the immune system not to attack the transplanted tissue could mean long-term acceptance of the transplanted organ.
Challenges to the clinical use of inverse vaccines
No inverse vaccine is currently available on the market, but clinical trials are in the early stages. Before they can be made available, certain challenges must be addressed:
- Accurately pinpointing the specific autoantigen (self-protein) responsible for each autoimmune disease is a major challenge. Some diseases might involve multiple autoantigens, making it trickier to design a targeted vaccine.
- Delivering the vaccine to the relevant antigen-presenting cells (APCs) and ensuring it reaches the targeted T cells involved in the problematic response is another hurdle.
- Achieving high specificity can be difficult — even slight variations in the autoantigen’s presentation could lead to unpredictable immune responses.
- Extensive clinical trials are needed to determine the safety and efficacy of inverse vaccines in humans, which can be lengthy and expensive.
- Due to the novelty of inverse vaccines, the long-term effects are unknown. More research is needed to understand their potential impact on the immune system over time.
While these hurdles are significant, the future of inverse vaccine research appears bright. With continued investment, scientific breakthroughs, and careful clinical evaluation, these novel vaccines have the potential to transform the treatment of autoimmune diseases and provide meaningful improvements in millions of lives.