Global Science & Technology Trends Report: Gene and Cell Therapy R&D
With rapid advances in recombinant DNA and protein engineering technologies, therapeutic fusion proteins have emerged as a new option for biologics with improved pharmacokinetic stability and enhanced capability to act on molecular targets. Fusion proteins have also facilitated the development of new vaccines, and gene and cell therapy drugs to address other clinical challenges.
Global Science & Technology Trends Report - Therapeutic Fusion Protein R&D
With rapid advances in recombinant DNA and protein engineering technologies, therapeutic fusion proteins have emerged as a new option for biologics with improved pharmacokinetic stability and enhanced capability to act on molecular targets. Fusion proteins have also facilitated the development of new vaccines, and gene and cell therapy drugs to address other clinical challenges.
CAS COVID-19 Protein Target Thesaurus
This open source dataset contains over 290K structure-activity relationship (SAR) data directed at COVID-19 protein, virus and disease targets reported in the literature, patents, and other public sources for over 9K of the substances included in the CAS COVID-19 Antiviral Candidate Compounds Dataset.
Your CAS COVID-19 Thesaurus Request
This open source dataset contains over 290K structure-activity relationship (SAR) data directed at COVID-19 protein, virus and disease targets reported in the literature, patents, and other public sources for over 9K of the substances included in the CAS COVID-19 Antiviral Candidate Compounds Dataset.
CAS COVID-19 Antiviral Candidate SAR Dataset
This open source dataset contains over 290K structure-activity relationship (SAR) data directed at COVID-19 protein, virus and disease targets reported in the literature, patents, and other public sources for over 9K of the substances included in the CAS COVID-19 Antiviral Candidate Compounds Dataset.
CAS COVID-19 Dataset Properties
The dataset is in SD file format (.sdf) and contains connection tables for nearly 50,000 chemical substances, along with related metadata such as CAS Registry Number® and physical properties for each substance.
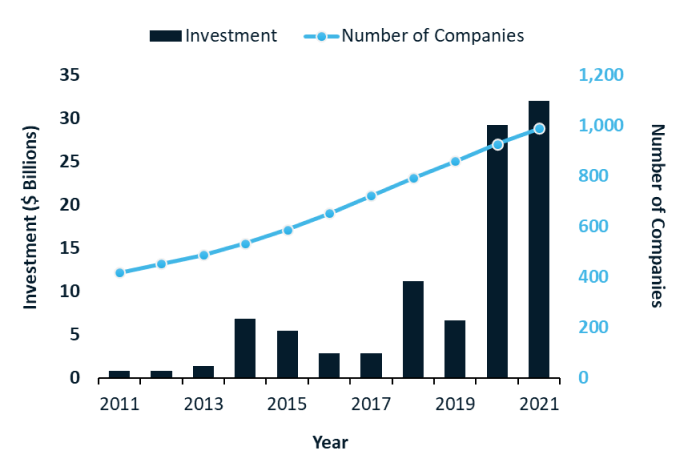
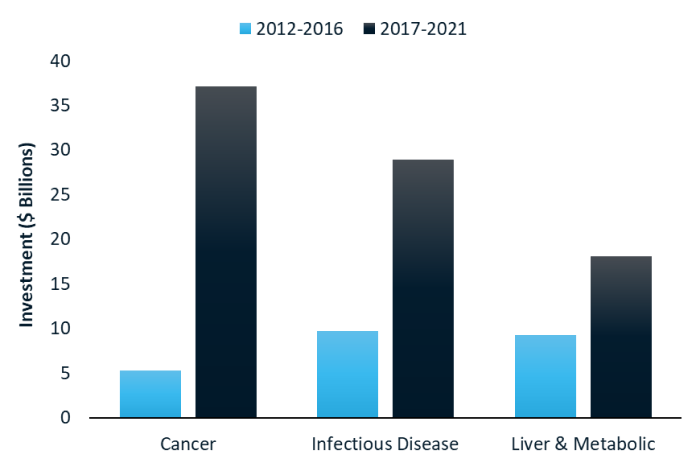
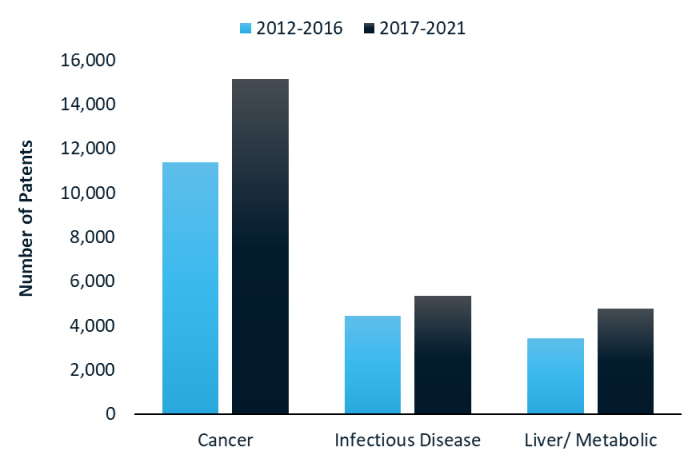
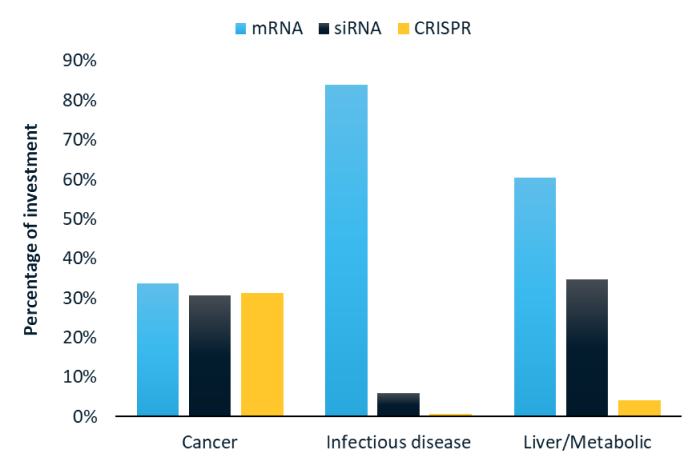
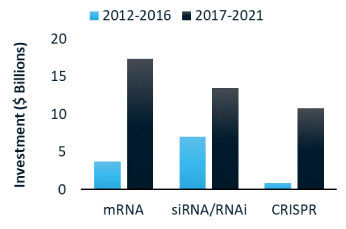
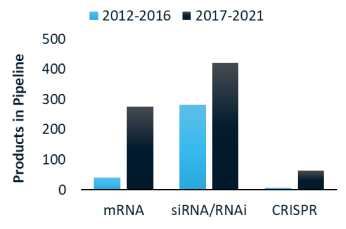
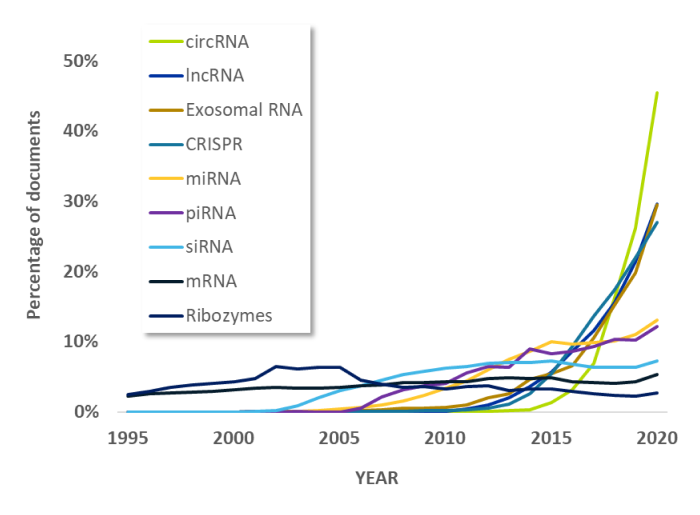
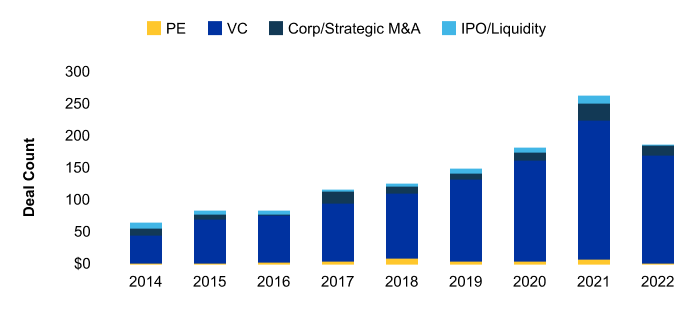
RNA medicines: what technologies are investors funding?
R&D Insights: exosomes in drug delivery and diagnostics