Gain new perspectives for faster progress directly to your inbox.

Exosomes are a nano-sized subset of extracellular vesicles released from cells as part of their normal physiology or under certain pathologies. In our previous blog on exosomal evolution, we discussed the advancement of these natural nanoparticles, from their initial discovery to the recent boom in extracellular vesicle research.
In part two of this three-blog series, we will explore further insights from the CAS Content Collection™, the largest human-curated collection of published scientific knowledge, summarizing the key applications of exosome therapy in drug delivery and diagnostics.
Rising research trends in exosome therapy
Using the CAS Content Collection, we analyzed the presence and trends of certain key concepts in scientific publications relevant to exosome applications in drug delivery and diagnostics (Figure 1). The keywords ‘targeting’ and ‘biomarker’ came out on top, reflecting the growing interest in exosomes in therapeutics. Significantly, analysis of the key concepts during the years 2017–2021 revealed a sharp increase in the term “blood-brain barrier” over the past two years, indicating that this is a hot topic in exosome therapy research. As we learned in part one, exosomes can cross the blood-brain barrier. The ability to cross this highly selective boundary not only makes exosomes valuable diagnostic tools, but it may also provide a means to deliver therapeutic cargo to the brain, helping to treat cancer and traumatic brain injuries.
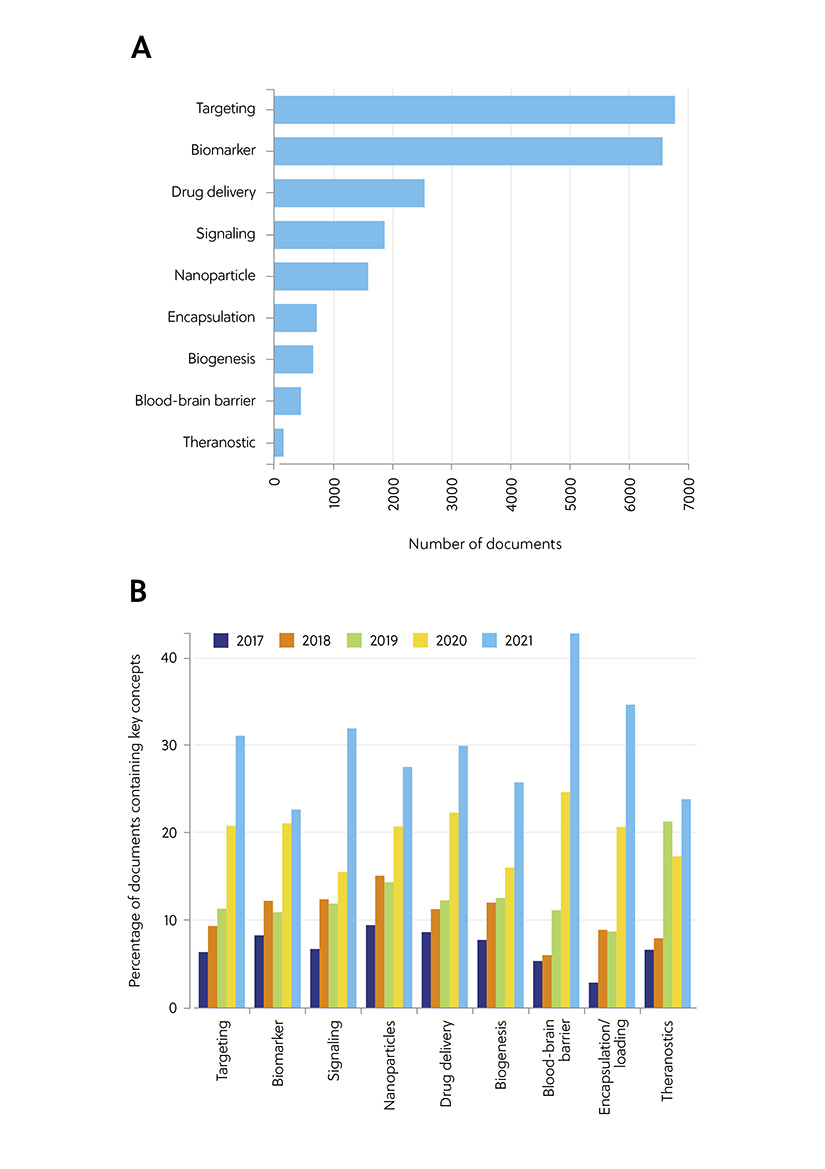
Figure 1. Key concepts in the scientific publications relevant to the exosome applications in drug delivery and diagnostics: (A) Number of publications exploring key concepts related to exosome applications in therapy and diagnostics. (B) Trends in key concepts presented in the articles related to exosome therapy applications and diagnostics during the years 2017−2021. Percentages are calculated with yearly publication numbers for each key concept, normalized by the total number of publications for the same concept in the same period.
The vital first step in exosome therapy: isolating and purifying exosomes
Before exosomes can be utilized in large-scale medical practice, it is crucial that these nano-sized particles are precisely distinguished from a wide spectrum of cellular debris and interfering components. There is no single standardized approach to exosome separation and analysis, with each approach providing unique strengths and limitations (summarized in Table 1). While ultracentrifugation was once considered the gold standard approach, in recent years precipitation and microfluidic methods have proved more popular due to their ability to purify exosomes without causing potential damage (Figure 2). A combination of several of these methods has been suggested as a promising strategy for the improvement of the isolation outcome. This is to provide exosome subsets with high purity with respect to size, morphology, concentration, the presence of exosome-enriched markers, and the lack of contaminants.
Table 1. Major methods of exosome isolation/purification
| Method | Advantages | Disadvantages |
| Ultrafiltration |
|
|
| Ultracentrifugation |
|
|
| Immunoaffinity |
|
|
| Polymer precipitation |
|
|
| Size-exclusion chromatography |
|
|
| Microfluidics |
|
|
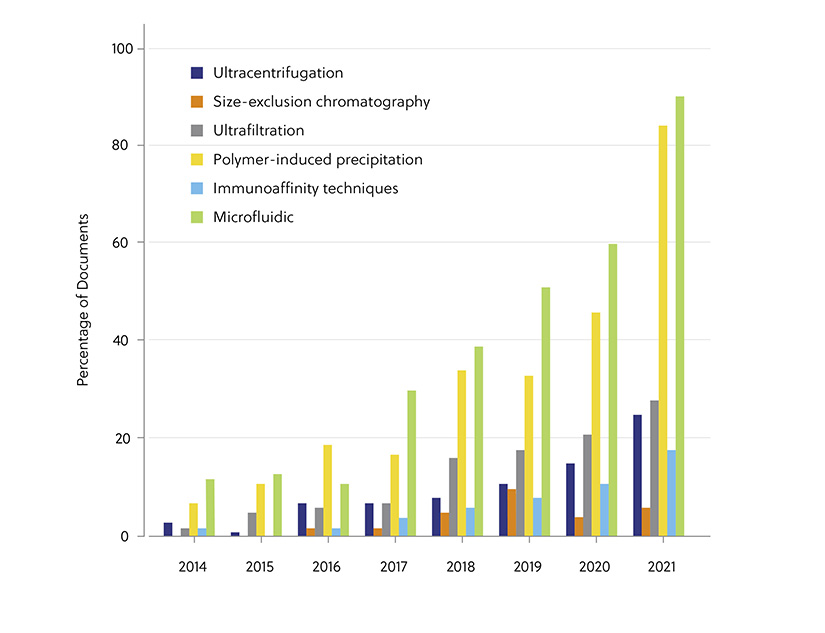
Figure 2. Trends in the number of documents related to exosome therapy applications and diagnostics concerning various exosome isolation methods during the years 2014−2021. (Percentages are calculated with yearly publication numbers for each isolation method, normalized by the total number of publications for the same isolation method in the same period.)
Exosome therapy and drug delivery
Once exosomes have been extracted and purified, how do we then turn them into effective drug delivery systems? Luckily exosomes are made for this role, combining the benefits of both synthetic nanocarriers and cell-mediated drug delivery systems, while avoiding their limitations. The first step to harnessing these properties is ‘cargo loading’, the process of packing exosomes with therapeutic materials. Several cargo loading methods have been employed for this purpose, each with their own advantages and disadvantages (Table 2).
Table 2. Cargo loading techniques
| Method | Advantages | Disadvantages |
| Cell transfection |
|
|
| Direct co-incubation |
|
|
| Sonication |
|
|
| Electroporation |
|
|
| Freeze-thaw |
|
|
| Extrusion |
|
|
As a form of cell-to-cell messengers, exosomes play a crucial role in different physiological processes. As such, exosomes secreted by different tissues and cells exhibit unique properties. For example, tumor-derived exosomes have been found to impact tumor properties such as growth, angiogenesis, invasion, and metastasis. In contrast, exosomes from mesenchymal stem cells (MSCs) have properties that make them ideal for use as adjuvants to support and complement other therapeutic modalities. In fact, a US-based company, Direct Biologics, is exploring the utility of the MSC-derived therapeutic ExoFlo in clinical trials for ulcerative colitis, solid organ rejection, and COVID-19, to name a few.
Though the potential applications of exosome therapy are wide-ranging, by far the most common area of exosomal research is cancer, followed by inflammation and infection. By analyzing the correlation between exosome donor cells and the diseases they have been applied to, a clear pattern emerges. Antigen-presenting cells and natural killer cells are most frequently used in cancer studies. Macrophages and stem cells are the most frequently used in inflammation, while antigen-presenting cells and T-cells are frequently used in infection (Figure 3).
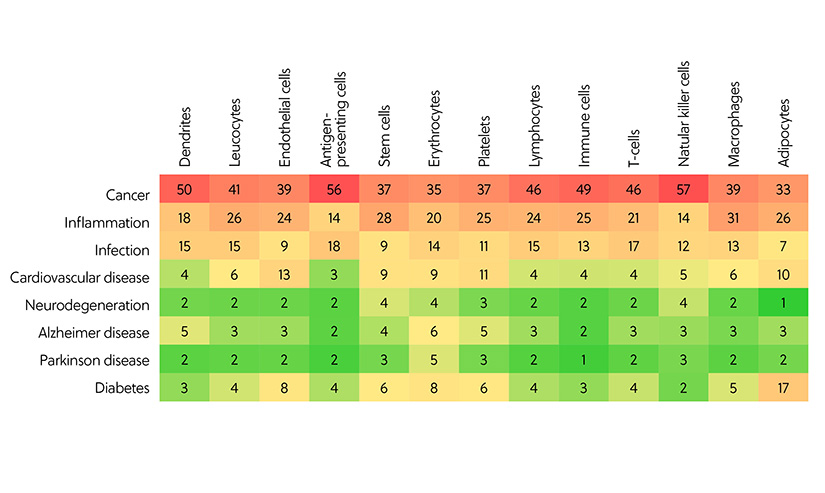
Figure 3. Correlation between exosome donor cells and the diseases to which the exosomes have been applied to in the studies related to exosome therapy and diagnostics, as represented by the number of documents in the CAS Content Collection.
The varied targeted applications of exosome therapy
Another rapidly expanding and noteworthy application of exosomes is their use as therapeutic agents. Exosome systems have been applied as therapeutic or diagnostic tools to a wide range of disorders. Our analysis of the CAS Content Collection shows that most publications (68%) on exosome therapy are associated with cancer. Exosomal microRNAs (miRNAs) have been shown to inhibit cancer cell proliferation, migration, and invasion. This approach has been explored in various malignant cell subtypes, including those for bladder, colorectal, and breast cancer. Exosomes also have enormous therapeutic potential in neurodegenerative, inflammatory, and cardiovascular diseases, which are also represented (Figure 4).
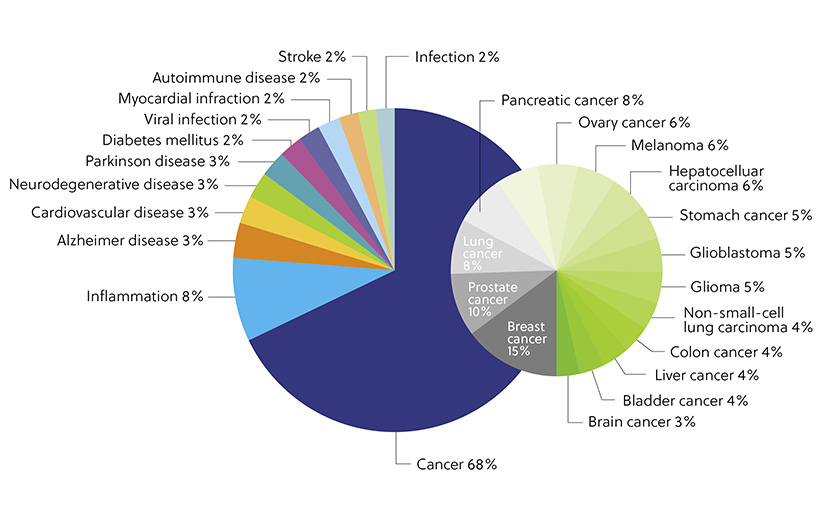
Figure 4. Distribution of the publications in the CAS Content Collection related to applications of exosome therapy and diagnostics with respect to the target diseases.
As exosomes are involved in the pathogenesis of diseases such as cancer, a successful therapeutic strategy may involve reducing elevated exosome production and circulation to normal levels to prevent disease progression. Several ongoing studies are exploring the impacts of modulating the exosome therapy pathway at various steps, including its production, release, and uptake. Physical elimination of exosomes has also been explored in cancer cells, with researchers hypothesizing that this elimination can hamper the communication between tumor cells that contribute to tumor progression.
Diagnostic use of exosomes
To be feasible for clinical use, a biomarker must exhibit several properties. It should be easy to access, cost effective, specific, highly sensitive, and measurable. Due to their unique properties, exosomes already tick several of these boxes, showing superiority over conventional serum-based biomarkers, particularly in regard to diagnostic sensitivity and accuracy.
There are several advantages to employing exosomes in this therapeutic way. Firstly, as the pathological status of cells greatly affects the content of exosomes (as observed in Alzheimer’s disease), studying these extracellular vesicles can provide a window into the disease state of the tissue. They are also innately stable, with a lipid bilayer that enables them to withstand degradation even within harsh microenvironments. In terms of practicality, exosomes can be easily and non-invasively isolated from biological fluids such as urine, blood, and even tears. Once extracted, they can be stored by freezing, freeze-drying, or spray-drying. Finally, unlike many conventional serum biomarkers, exosomes can pass through the blood-brain barrier, providing information about brain cells that would be otherwise difficult to obtain. Several candidate exosomal protein biomarkers (Table 3) and nucleic acid biomarkers (Table 4) are currently being explored. For the expanded list of these biomarkers see our ACS publication, Exosomes – Nature’s Lipid Nanoparticles, A Rising Star in Drug Delivery and Diagnostics.
Table 3. Examples of exosomal proteins for clinical diagnostic applications
| Proteins(s) | Disease | Body fluid |
| CD81 | Chronic hepatitis C | Blood plasma |
| CD63, caveolin-1, TYRP2, VLA-4, HSP70, HSP90 | Melanoma | Blood plasma |
| Epidermal growth factor receptor VIII | Glioblastoma | Blood plasma |
| Survivin | Prostate cancer | Blood plasma |
| c-src | Plasma cell dyscrasias | Blood plasma |
| NY-ESO-1 | Lung cancer | Blood plasma |
| PKG1, RALGAPA2, NFX1, TJP2 | Breast cancer | Blood plasma |
| Glypican-1 | Pancreatic cancer | Blood serum |
| Glypican-1 | Colorectal cancer | Blood plasma |
| AMPN VNN1, PIGR | Cholangiocarcinoma | Blood serum |
| CD24, EpCAM, CA-125 | Ovarian cancer | Blood plasma |
| CD91 | Lung cancer | Blood serum |
| Fetuin-A, ATF 3 | Acute kidney injury | Urine |
| CD26, CD81, S1c3A1, CD10 | Liver injury | Urine |
| NKCC2 | Bartter syndrome type 1 | Urine |
| EGF, α subunit of Gs, resisitin, retinoic acid-induced protein 3 | Bladder cancer | Urine |
| A2M, HPA, MUC5B, LGALS3BP, IGHA1, PIP, PKM1/M2, GAPDH | Squamous cell carcinoma | Saliva |
| LMP1, Galectin-9, BARF-1 | Nasopharyngeal cancer | Blood, saliva |
| CALML5, KRT6A, and S100P | Dry eye disease | Tears |
Table 4. Exosomal miRNAs as cancer therapeutic and diagnostic agents
| miRNAs | Cancer types | Applications |
| miR-378 | Non-small cell lung cancer | Prognostic |
| miR-423, miR-424, let7-i and miR-660 | Breast cancer | Diagnostic |
| miR-423-3p | Prostate cancer | Prognostic; castration-resistance |
| miR-30a | Oral squamous cell carcinoma | Therapeutic; cisplatin sensitivity |
| miR-106b-3p | Colorectal cancer | Therapeutic |
The interest in exosomes as biomarkers is reflected in the extensive growth in the number of documents related to applications of exosome therapy and diagnostics, as revealed by analysis of the CAS Content Collection (Figure 5). While it appears at first glance that documents related to therapeutics come out on top, the general percentage of documents is equally distributed between both applications.
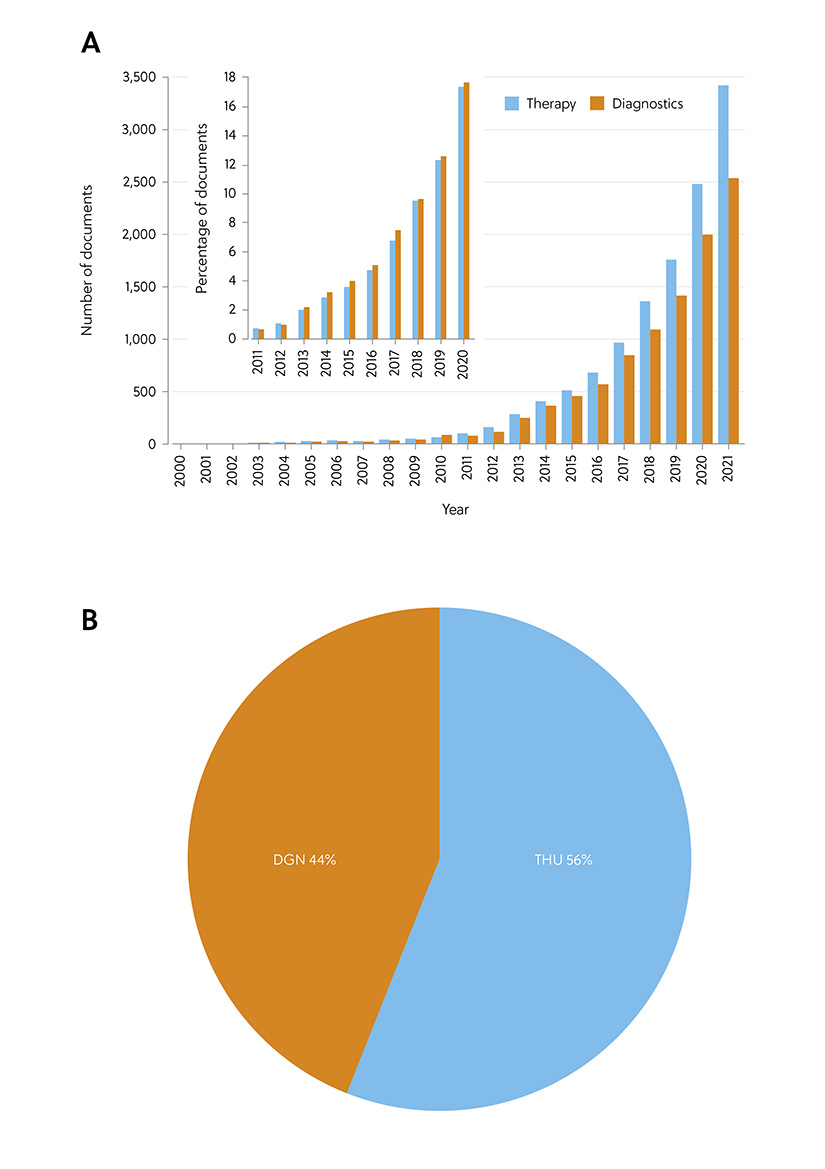
Figure 5. Diagnostic vs. therapeutic application of exosomes: (A) Comparison of the number of documents related to applications of exosome therapy vs. diagnostics; Inset: Annual growth of the number of documents related to exosome applications in therapy vs. diagnostics. (B) Comparison of the number of documents related to exosome applications in therapy vs. diagnostics with respect to their role indicators (THU, therapeutic; DGN, diagnostic).
While the current exosome therapy research is promising, many studies remain in the preclinical stage. That said, how close are we to utilizing the full potential of exosomes in therapeutics and diagnostics? What are the obstacles and challenges that stand in our way? In our final article in the series, we will uncover the key players in exosome research with updates on the key research initiatives in this exciting and dynamic field. In the meantime, you can read more in our Exosomes Insight Report.