Gain new perspectives for faster progress directly to your inbox.
As COVID-19 cases rise worldwide, the Omicron subvariant BA.5 is now the dominant strain sweeping across the US. The COVID BA.5 variant continues to mutate, increasing its spread and evading immunity. A recent pre-print study shows there are significant health risks with reinfections and new variants may be less affected by therapeutic monoclonal antibodies (mAbs).This blog explores key mutations that increase transmission, evade protective antibodies, and enable more reinfections.
Mutations increase the rate of infection
The spike protein is the key for entry into the human body and has been targeted and neutralized by many of the COVID vaccines. However, recent mutations by the spike protein from the Omicron variants (BA2.12.1, BA.4, and BA.5) suggest critical changes that enhance transmission.

As seen in Figure 1, these three new strains share key mutations that alter part of the receptor binding domain (RBD). This is the part of the spike protein that binds to the cells, enabling infection, and is also a key target for protective antibodies.
The F486V mutation found in BA.4 and BA.5 compromises the spike’s ability to bind to the viral receptor. The R493Q reversion mutation, however, restores receptor binding and therefore fitness.
Mutations increase resistance to treatment
While some mutations enable a faster spread of the virus, others enable the virus to be less affected by current therapies. Mutations to L452 may help the virus bind more closely to cells, hiding from disease fighting antibodies that try to block the virus. Researchers believe that this L452 mutation is the COVID-19 virus’s response to the huge Omicron surges of infection earlier this year. BA.4 and BA.5 also contain a mutation of the N-terminal domain (NTD) domain (Del69-70), which historically has also altered binding affinity, knocking out an antibody binding site. The F486V and R493Q mutations may also contribute to immune evasion from antibody binding, decreasing the efficacy of mAbs and ultimately increasing treatment resistance.
Bebtelovimab (CAS Registry Number: 2578319-11-4) is the only clinically authorized mAb to treat BA.2.12.1, BA.4, or BA.5 infections that has retained its potency against these newer subvariants. Bebtelovimab is a human immunoglobulin G-1 (variant) monoclonal antibody.
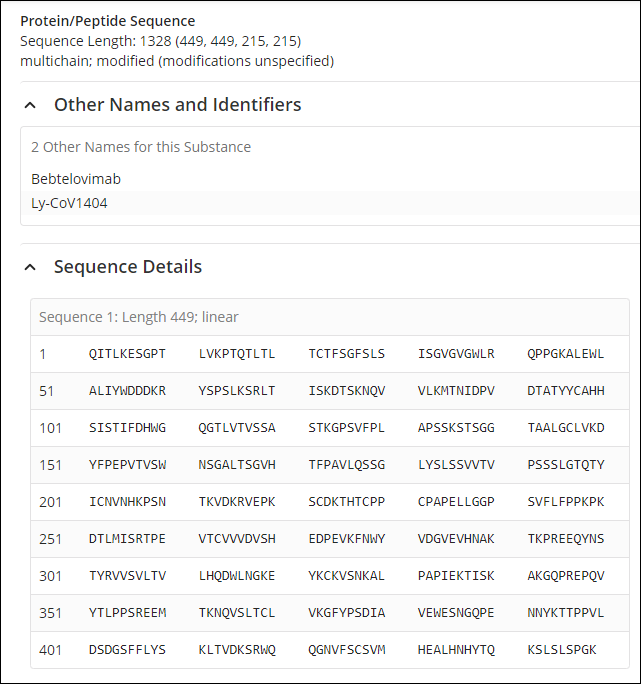
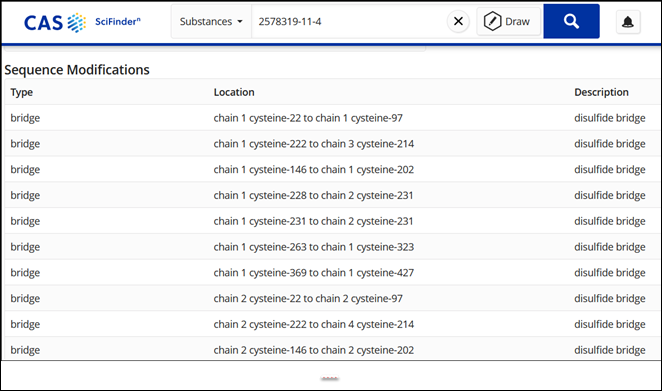
A CAS SciFinder substance search reveals that this therapeutic contains four protein sequences, two identical heavy chain polypeptides composed of 449 amino acids, and two identical light chain polypeptides composed of 215 amino acids. The sequences and modifications can be viewed in CAS SciFinder as shown in Figure 2 and Figure 3.
A recent article in the New England Journal of Medicine shows how the BA.2.12.1, BA.4, and BA.5 subvariants escape neutralizing antibodies induced by both vaccination and infection. They showed neutralizing antibody titers against the BA.4 or BA.5 subvariant and against the BA.2.12.1(lesser extent) subvariant were lower than titers against earlier BA.1 and BA.2 subvariants. These findings provide context for the current surges caused by the BA.2.12.1, BA.4, and BA.5 subvariants in populations with high frequencies of vaccination and previous infection.
Beyond the Spike Protein Mutations
While BA.4 and BA.5 are identical to each other in terms of spike mutations, they do share and differ in mutations that are outside of the spike protein. These mutations affect viral replication, infection rate, and treatment resistance. Both BA.4 and BA.5 revert two mutations back to the original virus, Orf6 D61 and NSP4 L438. Researchers believe that these mutations affect replication. Orf6 D61 resident protein Orf6 enhances viral replication by downregulating proteins, enzymes, and various signals. The NSP4 L438 resident protein NSP4 is involved in forming a double membrane vesicle, also potentially enhancing viral replication.
BA.4 has two mutations, L11F in Orf7B and P151S in the nucleocapsid (N) protein, the impact of which on antigen tests detecting the N protein is not yet established. Both the Orf7B and N protein mutations may contribute to immune evasion. The N protein mutation may also affect the stability of the virus, increasing fitness. BA.5 contains a D3N mutation in the membrane (M) protein - a relatively uncommon mutation. The M protein plays a role in suppression of immunity and also surrounds the virus, increasing possible cell invasion and transmissibility.
Vaccines proven effective against hospitalization
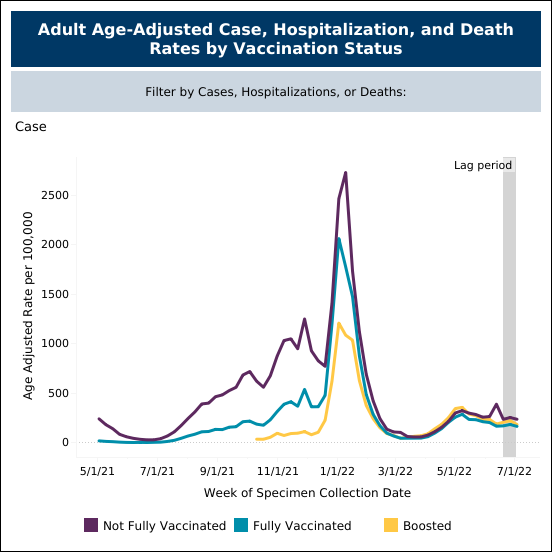
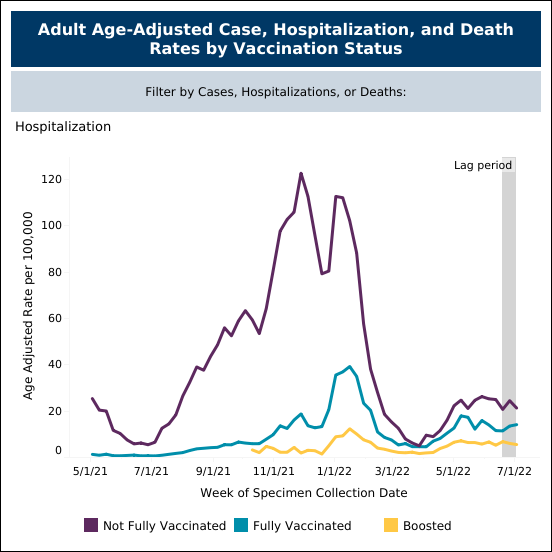
Current COVID-19 vaccines have minimal protection against symptomatic infections as seen in the data from Minnesota Department of Health, where fully vaccinated individuals are almost as likely to be infected as unvaccinated individuals (figure 4) in June and July when the BA.5 variant was rampant. However, when you examine hospitalization rates, the differences are dramatic as hospitalization numbers are substantially higher in the unvaccinated population (figure 5).
The current vaccines do offer good protection against severe symptoms, hospitalization, and death. The immunity generated by these vaccines help patients’ immune systems fight the virus and they are therefore less likely to develop severe symptoms and be hospitalized.
An article published in the New England Journal of Medicine discusses how current COVID vaccines are also effective against the BA.1 and BA.2 variants. This cross immunity has been recognized since the beginning of the pandemic. Evidence suggests influenza, measles, pneumonia and polio vaccines can all offer some level of protection against infection of SARS-CoV-2. According to the Mayo Clinic, people who received a pneumonia vaccine in the past year had a 28% reduced risk of COVID-19, while a 43% reduction in risk of COVID-19 infection was seen in people who received the polio vaccine.
Next Steps: Where do we go from here?
The COVID-19 pandemic is not slowing down as the virus continues to mutate and evolve across the globe. As shown above, current vaccines are still highly effective against severe disease, hospitalization, and death. However, COVID fatigue has prevented many from receiving their recommended booster vaccines. If an individual is eligible but has yet to receive a booster vaccine, they should receive one as quickly as possible. Renewed efforts to wear a high-quality mask when in confined, crowded environments will also help curb symptomatic infections.
Both Pfizer and Moderna have developed vaccines based on the new BA.1 variant that should be available in the US during the 2022 fall season. The limited protection that prior BA.1 infection provides against the newer variants raises questions about how useful this type of second- generation vaccine will be though. In the future, newer vaccine technologies combined with antiviral therapies that target a broader range of antigens will be required to stop the viral spread.